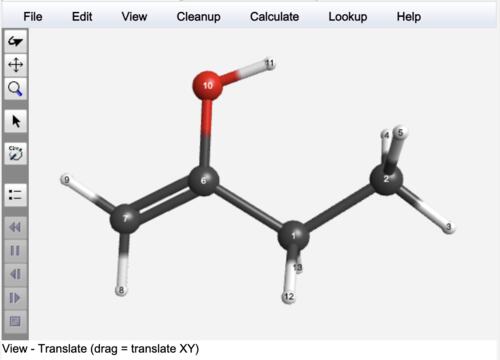
How to calculate ppm shift in webmo
NOTA and DOTA are used in many applications with their radiochemical properties ranging from sufficient to superb for many radiometals, including but not limited to 64Cu, 68Ga, 111In, 86/90Y, and 177Lu. (5, 7) The tri- and tetra-aza-based aminocarboxylate macrocyclic chelators 1,4,7-triazacyclononane-1,4,7-triacetic acid (NOTA (N 3O 3)) and 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA (N 4O 4)), developed by Maecke and co-workers, (16-18) persist as the “gold-standards” in the field of radiometal chelation. The utility of radiometals such as 111In and 68Ga in nuclear medicine is strongly dependent on a chelating ligand that exhibits rapid radiometal incorporation and strong kinetic inertness to prevent unintentional transchelation and/or demetalation of the coordination complex in vivo.
#How to calculate ppm shift in webmo generator#
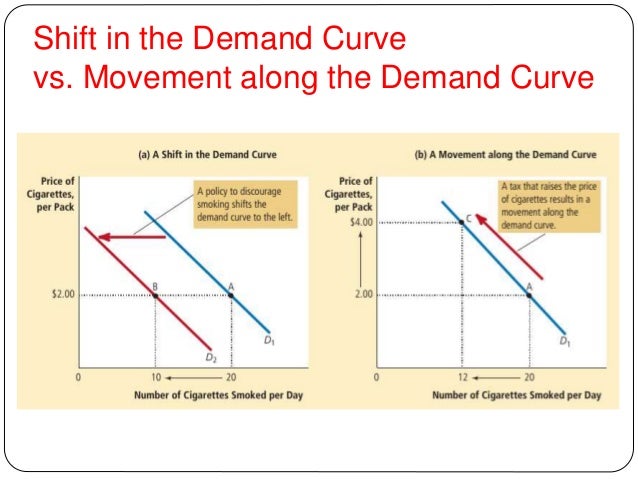
The lack of an FDA-approved 68Ge/ 68Ga generator is hindering the advancement of many 68Ga agents toward the clinic in North America, but progress is steady. Therefore, the 68Ge/ 68Ga generator has the potential to become as ubiquitous as the clinically important 99Mo/ 99mTc generator, and the 68Ga from these generators is already being used in patients throughout Europe. (1, 2, 5, 6, 12) Moreover, the nuclide 68Ga can be commercially produced and distributed via a 68Ge/ 68Ga generator system the half-life of the generator-parent 68Ge ( t 1/2 = 270 d) is sufficiently long such that the generator can be used for six months to one year before replacement, (13, 14) obviating the need for an on-site cyclotron. Initial investigations reveal that the chiral acyclic hexadentate H 2 CHXdedpa and octadentate H 4 CHXoctapa ligands are ideal candidates for radiopharmaceutical elaboration of gallium or indium isotopes, respectively.Ħ8Ga, with a short half-life suitable for imaging agents that localize quickly, such as small molecules and peptides ( t 1/2 = 67.7 min), and its predominant β + emission (89%, 1.9 MeV maximum energy), has become an attractive isotope for incorporation into positron-emission tomography (PET) imaging agents. The − complex showed exceptionally high in vitro kinetic inertness over 120 h in human serum, comparing well with previously reported − values, and an improved stability compared to the current industry “gold standards” 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) and diethylenetriaminepentaacetic acid (DTPA). Preliminary coordination studies of H 4 CHXoctapa with In(III) demonstrated − to have an equivalently high thermodynamically stable constant as −, with log K ML values of 27.16(9) and 26.76(14), respectively. In vitro kinetic inertness assays demonstrated the + complex to be more stable than + in a human serum competition, with 90.5% and 77.8% of 67Ga remaining chelate-bound after 2 h, respectively.
Like +, + exhibited exceptionally high thermodynamic stability constants (log K ML = 28.11(8)), and the chelate retained the ability to label 67Ga quantitatively in 10 min at room temperature at ligand concentrations of 1 × 10 –5 M. It was found that + showed very similar properties to those of +, with only one isomer in solution observed by NMR spectroscopy, and minimal structural changes in the solid-state X-ray structure. The ligands were compared to the previously studied hexadentate H 2dedpa and octadentate H 4octapa ligands to determine the effect adding a chiral 1 R,2 R- trans-cyclohexane to replace the ethylenediamine backbone would have on metal complex stability and radiolabeling kinetics.
The chiral acyclic ligands H 2 CHXdedpa (N 4O 2), H 2 CHXdedpa-bb (N 4O 2), and H 4 CHXoctapa (N 4O 4) ( CHX = cyclohexyl/cyclohexane, H 2dedpa = 1,2-methylamino]ethane, bb = N, N′-dibenzylated, H 4octapa = N, N′-bis(6-carboxy-2-pyridylmethyl)-ethylenediamine- N, N′-diacetic acid) were synthesized, complexed with Ga(III) and/or In(III), and evaluated for their potential as chelating agents in radiopharmaceutical applications.
